

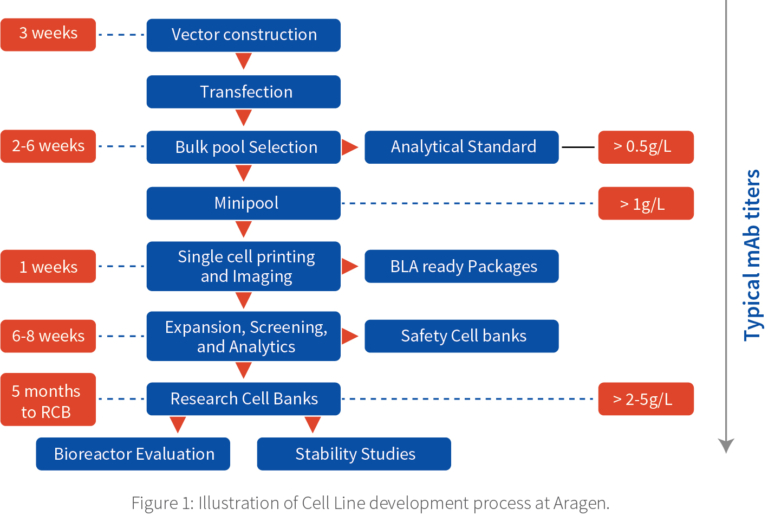
The cell line development (CLD) process starts with vector construction where our skilled molecular biologists perform in silico studies to do codon optimization and synthesis based on the sequence given followed by transfection in Aragen’s proprietary vectors.
Depending on the project, we do several transfections. The transfected cultures are incubated for 72 hours post which they are pooled together, called “bulk transfection.”
The bulk pools are seeded on T75 cell culture flasks and are purified after two weeks. Bulkpool is used for a fed-batch production run to generate materials that is purified and analysed as an analytical standard or benchmark against the expected product quality.
To identify the best transfected cells or hot spots the bulk transfection is divided into several tiny populations called minipools and the leftover is referred as the bulkpool.
Minipools help identify the high producing cells. The minipools are plated in 96 well plates and later they are screened in 24 well stage, after which the top 20 minipools are identified and are evaluated in shaker flask. It takes around six weeks to complete the selection process.
After shaker flask evaluation, the top performing clones are taken for single cell printing and imaging. Based on the titer data, top few mini-pools are plated for single cell printing, using a microfluidic-based platform, CYTENA, which ensures that each well has only one cell. Cells are left to divide for 3–4 days, and the cell population is confirmed using the Solentim imager.
Post the shaker flask evaluation, the protein is purified, and the analytics and characterization are performed to calculate parameters like cell viability, metabolic profile, titer, and quality of protein and three clones are selected for the research cell banking (RCB).
The top 20 cell clones which are fully adopted in the shaker flask are banked in three vials each and they are referred as safety cell bank.
For manufacturing the proteins in large quantities, we use bioreactor evaluation where all the parameters like temperature, pH, dissolved oxygen, agitation can be controlled. We start with 1L bioreactor and is scalable to 2L, 5L, 10L, 50L to 2000L. The selected RCB undergoes
stability studies for 60 to 100 generations. The total time taken to achieve the RCBs is around 5 months.
In cases where the timelines are challenging, the process development in bioreactors can be conducted with parent minipools. This way, the process
development, and MCB generation move forward in parallel, cutting the timelines by around two months. Cell line stability studies can also be run in parallel with master cell bank generation using interim clones, further reducing the timelines.
Aragen has over two decades experience of successfully delivering integrated and standalone solutions in research, optimization, and development of biological solutions. Our depth of expertise enables us to produce novel cell lines with maximum yield, while reducing processing time. We believe that every minute of our clients’ and, more significantly, every minute of our patients’ unmet medical requirements is crucial.
Our decades of experience on more than 120 biomolecules and engineering numerous cell lines makes us one of the most sought partners in life sciences.
